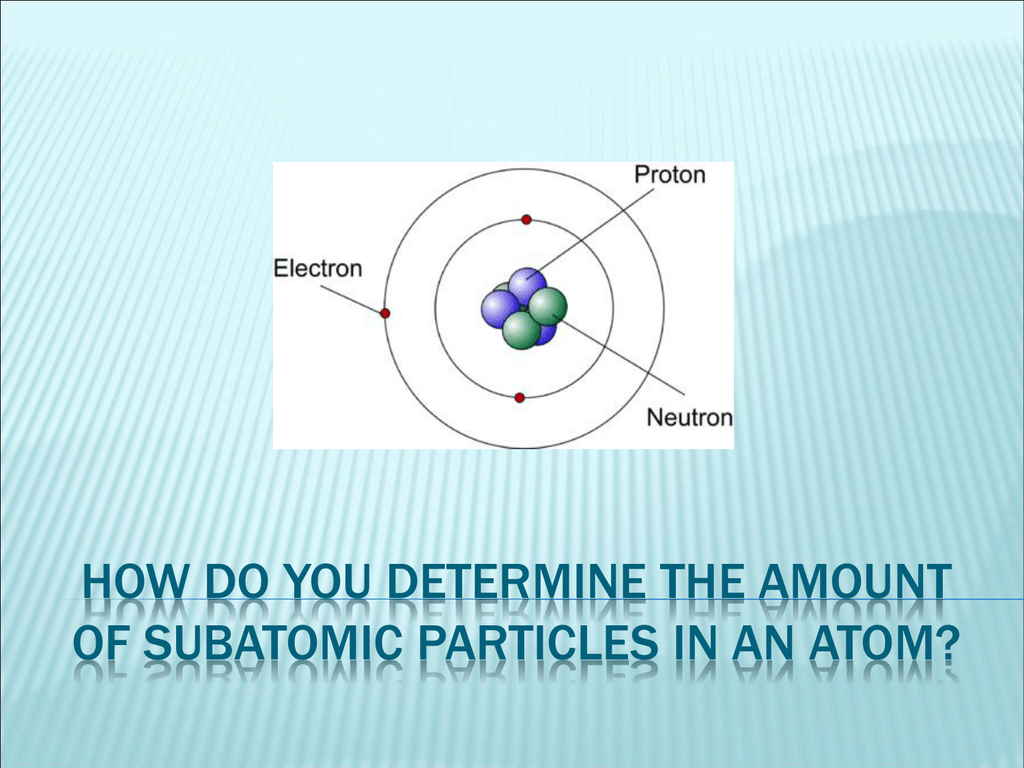
Atoms are made of protons, neutrons, and electrons, which are types of particle also known as subatomic. **Using relative charges and masses are used to keep calculations simple. Atoms consist of protons, neutrons and electrons. :max_bytes(150000):strip_icc()/atom-model-close-up-soft-focus-toned-b-w-AA038275-584ee71d5f9b58a8cd2fd2e8.jpg) 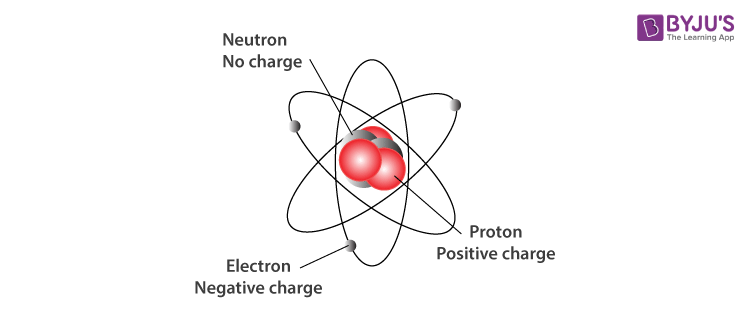
They have the same atomic number (same number of protons and electrons), but a different mass number due to more or fewer neutrons. Isotope is one of two or more forms of atoms of an element that differ in their number of neutrons. For a given element, the mass number is the number of protons and neutrons (nucleons) in the nucleus. Answer: Solution : Allied Solutions - Simplified Middle School Chemistry - Class 8 chapter Atomic. Neutron is an uncharged particle of the nucleus of all atoms EXCEPT hydrogen. The first is the tiny atomic nucleus, which is in the center of the atom and contains positively charged particles called protons and neutral, uncharged. State where the subatomic particles are present in the atom. Ion is an atom that has gained or lost one or more electrons, thus becoming positively or negatively charged. An atom is made up of about 200 different types of particles such as proton, neutron, electron, muon, positron, tau, and blah blah blah. Electrons can be shared or transferred among atoms.Ītoms have an equal number of protons and electrons therefore, they have a no net charge. any Chlorine atom is going to have 17 electrons). That theory, put forward by physicist Murray Gell-Mann in 1964, describes how the protons and neutrons that make up atomic nuclei are themselves composed of. Protons and neutrons are in the center of the atom, making up the nucleus. All atoms of an element have the same number of electrons (i.e. Atoms are made of extremely tiny particles called protons, neutrons, and electrons. The atomic number of an element represents the number of protons in the nucleus.Įlectron (e -) is negatively charged particle that can occupy a volume of space (orbital) around an atomic nucleus. Proton (p +) is positively charged particle of the atomic nucleus.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
